- Home
- About
-
9th Grade
- Rube Goldberg
- Physics of Sports Video
- Fire Away and Hybrid Car
- Creative Engineering Design
- Passive Solar Design - Design our new San Marin High School STEM building
- Satellite - Put something in Orbit
- Build a Musical Band - waves
- Robot Art Show - electricity, electronics, and programming
- Do It Better - Engineering Redesign
- 10th Grade
- 11th Grade
- PE
Project goal and Proof of Work
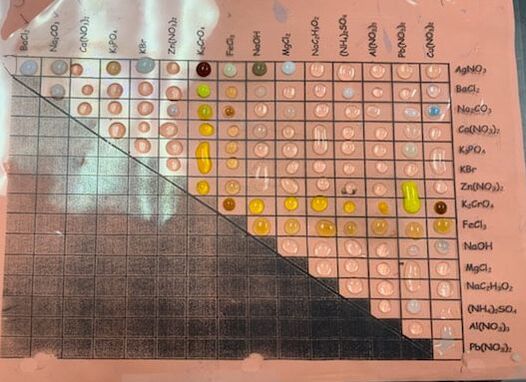
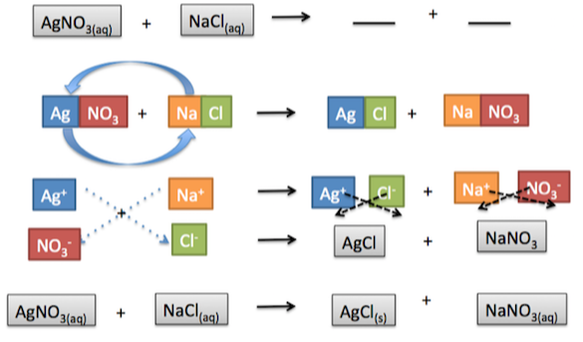
Our goal for this project was to use our knowledge of chemistry (types of reactions, and what the products typically of those reactions) to create art. My group proposed making our own paints by combining the correct chemicals to get the pigment we wanted. We first made a template of the San Marin emblem to trace onto our canvas, I then needed my color and did so by combining the chemicals sodium carbonate (Na2Co3) and copper(ll) sulfate (CuSo4). A double displacement reaction occurs and a pigment is left behind. I then filtered all the pigment and left if out to dry. You can see below is a chart of some double displacement reactions that we did before hand to try and discover patterns to help us create our pigment for our paint. We also had a general rule of which combinations of chemicals would produce the reaction we were looking for or not which helped guide us. Next I had to mix the chalky dye we made into a paint base and created the paint you see on our art. Mine is the white and blue emblem and you can can see that it wasn't perfect but the blue came out pretty well.
Reflection:
This project began pretty difficult for me but as I spent more time thinking about it and working at it, I was able to better wrap my head around it and create the dye you saw with chemistry and work that I did, which is a super satisfying feeling. Our group worked very well together but we could get distracted at times so for my future projects, I want to focus on my work ethic regardless of what my group is or isn't doing. Funny enough though, I believe my work ethic and drive was much higher on this project because I knew I needed to understand it well to have any chance at having a good looking final piece. I just need to apply myself correctly and I can be productive.